Percent Ionic Characater Calculator This spreadsheet will compute the percent ionic character of a covalent bond using the Pauling equation. Ionic character 1823984 100 4568 When electronegativity difference between two atoms is 21 there is 50 ionic character in the bond.

Calculate The Percentage Of Ionic Character In Cs Cl Bond In Cscl Molecule The Electronegativity Values Of Cs And Cl Are 0 7 And 0 3 Respectively
Using the molecular or bond dipole moment to calculate the percent ionic character plus an example.

How to calculate percent ionic character. Enter the symbols of the elements forming the bond in the appropriate cells. Bonds with more than 50 ionic character are considered ionic. The percentage of ionic character in a compound having some covalent character can be calculated by the following equation.
Every ionic compound having some percentage of covalent character according to Fajans rule. The spreadsheet will look up the electronegativities and do the calculations. Percent ionic character calculator uses percent_ionic_character 100 1-exp-025 Electronegativity of element A-Electronegativity of element B2 to calculate the Percent Ionic Character Percent ionic character is a measure of compound or molecules ionic and covalent character.
A bonds percent ionic character is the amount of electron sharing between two atoms. What I know is that the percentage ionic character is observed dipole moment divided by calculated dipole moment 100. According to Pauling when the electronegativity difference between two elements is 17 percentage of ionic character is 50 ie.
The calculated dipole moment is charge on electron. The percent ionic character Observed dipole momentCalculated dipole moment assuming 100 ionic bond 100. Materials science and engineering tutorial.
Percent ionic character 1 e 2 2 100. In HF the electronegativity difference between H and F is 19. If two atoms A and B are forming a covalent bond A-B then Percentage of ionic character.
Calculate The Percentage Ionic Character Of The Semiconducting Compounds CaAs And ZnSe Using Paulings Equation. IC100 1e EN 4. For a given bond the percent ionic character is given simply by Here I is the percent ionic character m obs is the actual observed or in the case of bond dipole moments calculated dipole moment and m ionic is the dipole moment which would occur if the bond were 100 ionic.
M ionic is easily calculated. Therefore ionic character is computed to be 56. To determine a bonds percent ionic character the atoms electronegativities are used to predict the electron sharing between the atoms.
Limited electron sharing corresponds with a high percent ionic character. The percentage ionic character in covalent compound arises due to the electronegative difference of bonding atoms. And percentage ionic character of covalent bond calculates.
We can calculate the ionic character of any molecule if we know the electronegativity values of both the atoms which are sharing an electron cloud sharing an electron cloud forming a covalent bond by using the following formula. 10000 9000 8000 Percent ionic character. When electronegativity difference is zero identical atoms the bond will be 100 covalent.
Worked example problem solution for percent ionic character calculation. But Id like to correct the definition of percent ionic character in your question using dipole moment not Observed value of ionic character. 100 Interatomic spacing electron charge100 0156109 161019 C mUsing 1D 3381030 100 1615633 756D ioniccharacter 100 observed 844.
EN ionic character 100 1 - e -EN4 Using this equation the percent ionic character of NaCl bonds is about 70 and the percent covalent character is. Pauling developed an empirical formula for approximating the percent ionic character for a bond based on the electronegativity difference. Whether a compound is polar covalent or ionic can be determined by calculating the percent ionic character which is the ratio of a bonds measured dipole moment to the dipole moment assuming a complete electron transfer.
Percent ionic character observed calculated 100.

The Percentage Ionic Character In H Br Bond In Hbr Molecule Is Youtube
Calculation Of Percentage Ionic Character
Calculation Of Percentage Ionic Character

Calculate The Percentage Of Ionic Character In K Cl Bond In Kcl Molecule The Electronegativity Values Of K And Cl Are 0 8 And 3 0 Respectively
Calculation Of Percentage Ionic Character

What Is The Chemistry Questions
C Calculation 30 11 Compute The Percent Ionic Chegg Com

Estimate The Percent Ionic Character Of The Hbr Molecule Given That The Dipole Moment Mu Is 0 63d And Hbr Bond Length 187 5 Pm Sahay Lms

Percent Ionic Character Worked Example Problem Youtube

Trick To Solve Percentage Ionic Character Problems Easily From Dipolemoment Youtube

Estimate The Percent Ionic Character Of Th Clutch Prep

Calculate The Percentage Of Ionic Character Of Hf Given That The Dipole Moment Of Hf Is 1 91 D And Its Bonding Length Is 0 92a
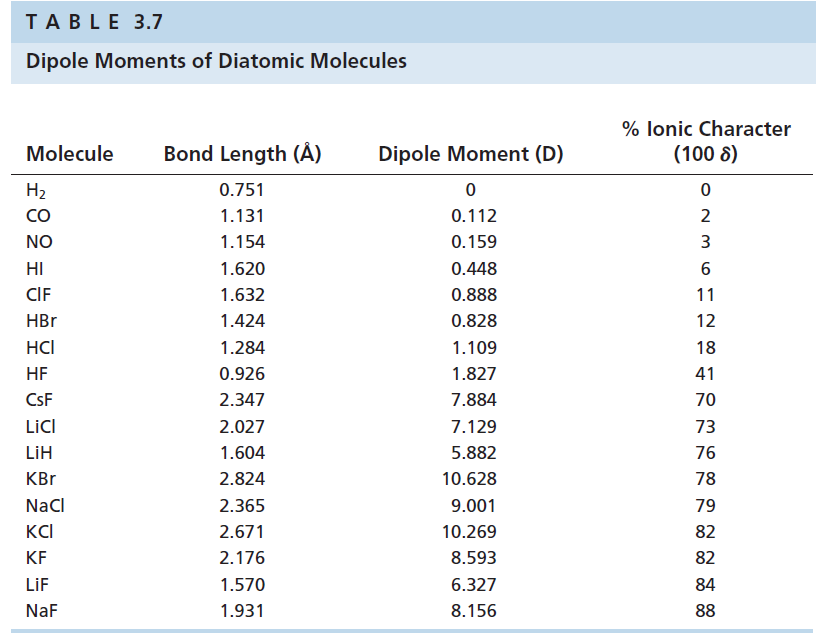
Solved The Percent Ionic Character Of The Bonds In Several Interh Chegg Com

The Electronegativity Difference Between Two Atoms A And B Is 2 Then Percentage Of Covalent Youtube

Calculate The Ionic Character For Molecule Ab When E N Different Is 2 0

Estimate The Percent Ionic Character Of The Hbr Molecule Given That The Dipole Moment Mu Is 0 63 D And Hbr Bond Length 187 5 Pm Sahay Lms






